Hypoglossal Nerve Stimulation: A CPAP User Looks at the Surgical Alternative
Every few weeks, someone emails me asking about hypoglossal nerve stimulation. Usually it is someone who has been fighting their CPAP for months or years, and they want to know whether the implant they keep seeing on Facebook ads is the way out. The question got more interesting in August 2025, when the FDA approved a second device, the Nyxoah Genio. For more than a decade, Inspire was the only option. Now there are two.
I should say up front: I have not had this surgery. My background is in computer science, not medicine. I was diagnosed with severe obstructive sleep apnea more than a decade ago at an AHI of 51, and I have managed it since with a ResMed AirSense 10 and a full face mask. CPAP works for me. So this article is not me telling you what it feels like to live with an implanted nerve stimulator. It is me, a long term CPAP user, reading the research on a treatment I keep getting asked about, and writing down what I found.
Any actual decision belongs in a conversation with a board-certified sleep doctor and an otolaryngologist who does these surgeries regularly. Not with me, and not with a blog post.
What the device actually does
If you have been on CPAP, you already know the basic problem. When you fall asleep, the muscles in your upper airway relax. The tongue base falls back, the airway closes, you stop breathing, your brain panics you awake just enough to fix it, and the cycle repeats all night. CPAP solves this by blowing air down the airway hard enough to keep it open from the inside.
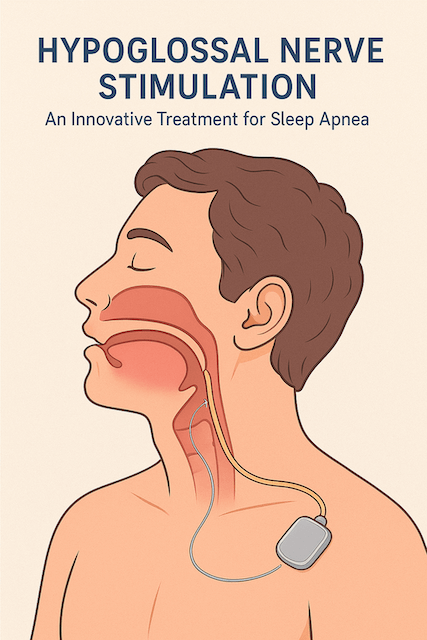
Hypoglossal nerve stimulation solves it from a completely different direction. Instead of pushing air, it pokes the nerve that controls your tongue. A small electrical pulse, timed to your inhale, contracts the tongue muscle and nudges it forward just enough to keep the airway open. No mask, no hose, no machine on the nightstand. A surgically implanted device, a remote control on your bedside table, and a button you press before sleep.
The first device, Inspire, has three pieces. A pulse generator the size of a pacemaker sits under the skin in the upper chest. A stimulation lead wraps around the part of the hypoglossal nerve that pushes the tongue forward. A pressure sensor sits between the ribs and tells the generator when you are inhaling so the pulse arrives at the right moment.
The newer Genio is built differently. The implant goes in the chin through a single incision. There is no internal battery and no separate breathing sensor. A wearable chip that sticks under the chin at bedtime powers the implant wirelessly and provides the timing. Genio also stimulates both sides of the tongue rather than just one. Different engineering, same goal.
How we got here
The story really starts with the STAR trial, published in the New England Journal of Medicine in 2014. Researchers implanted 126 patients with moderate to severe obstructive sleep apnea who had not been able to make CPAP work. After twelve months, about two thirds of them had cut their AHI by more than half and gotten it under 20. The median AHI dropped from around 29 to 9. People felt less tired. Their sleep questionnaires improved. The FDA approved Inspire that April.
What I found more interesting than the original trial is what happened next. Five years later, researchers caught up with the patients who were willing to come back, and roughly three quarters of them were still doing well. The serious complication rate stayed low, and most of those complications were lead adjustments rather than nerve damage. For a brand new implanted device, that is a reassuring number.
Then real world centers started reporting their own results, and they often looked even better than the trial. The team at Cleveland Clinic published a piece on their first batch of patients showing average AHI dropping from the mid-40s to under 5. They argued that part of the reason post-trial outcomes look stronger is that surgeons have refined the procedure (it has gone from three incisions to two), patient selection is tighter, and the team programming the device knows what they are doing in a way that early adopters did not.
So when Genio finally arrived in August 2025, it joined a therapy with more than ten years of follow-up data and several thousand patients implanted. That matters. Sleep apnea patients have a healthy skepticism of “innovative new treatments,” and rightly so. This one has earned the time on the field.
Who actually qualifies
This is where the conversation usually gets practical. Not everyone with sleep apnea is a candidate, and the rules are stricter than for CPAP.
The basics: AHI between 15 and 65 events per hour. BMI of 32 or less, with some labels stretching that to 35 with a warning. CPAP must have been tried and either failed to work or proven impossible to tolerate. Central sleep apnea disqualifies you, because the implant treats the obstructive kind, not the kind where your brain forgets to send the breathe signal. And you need to be an adult.
The most important screening step happens in an operating room before the implant is even planned. It is called drug-induced sleep endoscopy, or DISE for short. The surgeon sedates you with propofol to mimic real sleep, then runs a flexible scope through your nose and watches how your airway collapses. Two patterns matter. If your soft palate falls backward against your tongue base (called anterior-posterior collapse), the implant tends to work well. If your airway collapses from all sides at once like a closing drawstring (concentric collapse), it tends not to. The FDA labeling for Inspire excludes that second pattern. Genio, interestingly, has a separate program in Europe specifically for those patients, and that may eventually open up the therapy to people who are currently turned away.
If you are getting evaluated, expect DISE to be required, not optional. Medicare and most private insurers will not cover the implant without it.
I want to flag something that came up over and over in the research. CPAP failure has to be real, documented, and worked. Different masks tried. Pressure adjusted. BiPAP attempted where appropriate. Insurance companies will look at this carefully, and so will the surgical team, because CPAP that you can actually wear reduces AHI more aggressively than HNS does. CPAP is still the first line treatment, and that is not corporate caution, it is what the data say. The implant is for the people who genuinely cannot make CPAP work, not for people who would rather not.
What the long term data look like
Here is what I find encouraging when I read the research. A 2022 review in the Journal of Clinical Sleep Medicine pulled together post-market surveillance data on more than 20,000 patients. The one year explant rate (people having the device removed) was under 1 percent. The one year revision rate (going back in to fix something) was around 1.5 percent. By three years, those numbers crept up to roughly 3.6 and 7.1 percent. Those are reasonable numbers for an implanted device used for a chronic condition.
The number that gets attention, though, is adherence. CPAP, depending on whose data you trust, has long term adherence somewhere in the 30 to 60 percent range. People stop using it. They get tired of the mask, the hose, the noise, the weight on their face, the dry mouth, the leaks. HNS adherence is much higher, often quoted in the 80 to 90 percent range. There is no mask to put on, just a button to press, and patients who get past the adjustment period tend to stay with it.
That gap matters more than the per-night efficacy gap. CPAP, used every night with a sealed mask, can drive AHI under 5 for most patients. HNS more typically lands in the high single digits or low teens. CPAP wins per night when worn. But CPAP that is sitting unused in a closet does nothing for you. The honest comparison is between CPAP as people actually use it and HNS as people actually use it, and that is a much closer race than the trial numbers suggest.
The honest risks
This is where I think a lot of marketing material undersells things, so let me be straight about what I read.
It is surgery. Real surgery. The version most centers do today is two incisions for Inspire, one for Genio, but you are still going under general anesthesia, you still have implanted hardware, and you still have a recovery period. Infection rates are low (under 2 percent), but not zero. Bruising, swelling, and incision pain are normal for the first few weeks.
The thing that surprised me when I read patient reports is how long the adjustment period is. The device is usually activated about a month after surgery, once swelling has settled. Then the team gradually increases the stimulation over weeks, often with a titration sleep study to dial in the right settings. Tongue discomfort during stimulation is the most common complaint, and most people get used to it after two or three months. People going in expecting an instant cure tend to be disappointed. People going in expecting a slow ramp tend to do well.
Battery replacement is a thing for Inspire, eventually. The current generators are rated for somewhere in the seven to eleven year range, and when the battery runs down, you go back in for a smaller surgery to swap the generator. Genio’s external battery sidesteps this entirely, which is one of the bigger practical arguments for it.
MRI is worth flagging if you have other health issues. Inspire is MRI-conditional, meaning you can have a scan but only with specific protocols. Genio is fully MRI compatible at both common scanner strengths. If you are someone who is likely to need imaging in the future, this is a real conversation to have with your doctor.
And about a quarter to a third of patients in the trials did not hit the treatment success threshold. The implant is not a guarantee. It works very well for most appropriately selected patients, and it does not work for everyone.
The wallet conversation
HNS is expensive. The total cost of the procedure, depending on which study you read and how the contracts are written, lands somewhere in the range of 30,000 to 50,000 U.S. dollars. Medicare and most major private insurers cover it, but only if you can document that you meet all the criteria: confirmed sleep study, CPAP failure, BMI within range, favorable DISE result, letter of medical necessity from a sleep physician. Prior authorization is the rule.
What you actually pay out of pocket depends entirely on your insurance plan. Deductibles, coinsurance percentages, out of pocket maximums. People I have talked to in the U.S. report wide ranges, from a few hundred dollars to several thousand, depending on where they are in their plan year and what kind of coverage they have.
The cost-effectiveness research generally shows HNS is reasonable on the standard health-economics measures, though more expensive than CPAP. That math shifts a lot if the alternative is no therapy at all because CPAP simply does not work for the patient. Untreated severe sleep apnea is also expensive. It just shows up in different parts of the medical bill.
CPAP versus the implant, in practical terms
Here is the comparison I would lay out for a friend who was weighing this. The numbers come from the published research; the framing is mine.
| Factor | CPAP | Hypoglossal Nerve Stimulation |
|---|---|---|
| AHI reduction when used | Usually 90 percent or better | Usually 65 to 75 percent |
| Adherence in the real world | Roughly 30 to 60 percent | Roughly 80 to 90 percent |
| External equipment at night | Mask, hose, machine | None |
| Surgery | None | Outpatient implant procedure |
| Cost up front | Hundreds to a few thousand, plus supplies | Roughly 30,000 to 50,000, often largely covered |
| Time to full benefit | A few weeks | Two to three months of titration |
| Stopping treatment | Just stop | Requires explant surgery |
If I had to summarize, CPAP is more effective per night, much cheaper, and easy to walk away from. HNS is harder to undo once implanted but tends to actually get used. For someone who is going to use their CPAP, CPAP is the better answer. For someone who has genuinely tried and cannot get there, the implant becomes a serious option.
Where the field is heading
A few things worth keeping an eye on if you are following this space.
Studies in adolescents and young adults with Down syndrome (a population with very high rates of severe sleep apnea and a lot of CPAP intolerance) have been encouraging, with consistent AHI reductions and improvements in daytime function. FDA approval for that specific group is widely expected.
Several centers are publishing outcomes in patients who fall outside the original criteria, including BMI somewhat above 32. The picture is mixed, but the trend is toward broader eligibility.
The Genio program for patients with concentric collapse, who are currently excluded from Inspire, could meaningfully expand who is eligible. The ongoing ACCCESS trial in the U.S. will tell us more.
And on the boring-but-important side, adherence monitoring on Inspire is now cloud-based, which gives clinicians the same kind of objective use data that CPAP machines have been streaming for years. That is going to change how the therapy is managed over time.
Questions worth bringing to a real consultation
If you are seriously considering this, the appointment with the sleep physician and the surgeon is more useful than any article. A few things I would want to ask, written down before I went in.
How many of these implants has the surgeon personally done, and what are the typical outcomes at that center? What does my insurance actually cover and what is my realistic out of pocket exposure? Will I need a DISE before any of this can be answered, and what does that cost? What does the activation and titration schedule look like, and how many follow-up appointments should I plan for in the first year? What happens if my response is not what we hoped for? Are there CPAP alternatives I have not actually exhausted yet, like different masks or BiPAP?
Centers and surgeons vary. Volume matters in surgery. The honest, direct answers to those questions will tell you whether you have found a strong program.
My honest take
I will be straight with you. I have been on a CPAP for more than a decade. The mask is part of my life. I have camped with it. I have flown with it across hemispheres. I am even thinking about an upgrade to the AirSense 11. CPAP is not glamorous, but for me, it works.
For people CPAP works for, CPAP is the right answer. It is cheaper, it is reversible, and the data still favor it on a per-night basis. The marketing around HNS sometimes glosses over that, and I think that is a disservice.
For people CPAP genuinely does not work for, after a real, documented, exhausted effort, hypoglossal nerve stimulation is a real option backed by serious clinical evidence. The original device has more than ten years of data behind it. A second device is now on the U.S. market. The therapy is no longer experimental in any meaningful sense. It is mainstream sleep medicine.
But it is also a permanent surgical implant in your chest or chin, with an adjustment period that takes months, a price tag that depends on your insurance, and a roughly one in three chance of not being the answer you hoped for. That is a real decision, and it deserves a real conversation with people who do this for a living.
If you are weighing it, my advice is the same advice I would give a friend. Make sure you have actually tried the CPAP options that exist, including different masks and BiPAP if your sleep doctor thinks it is worth a shot. Get evaluated by a sleep medicine center that runs a serious HNS program. Insist on understanding the DISE result and what it means for your odds. Ask about cost. Talk to patients on both sides of the procedure if you can. And remember that the goal here is not the device. The goal is sleep, and the breathing that goes with it.
If you have questions or you have been through this yourself, I read every comment. The next person reading this might really benefit from hearing what your experience was actually like.
A few good places to read further
For background on what AHI actually measures, see my page on the Apnea-Hypopnea Index. The Epworth Sleepiness Scale explains the daytime sleepiness measure that keeps showing up in these trials. If you are weighing surgical options more broadly, my pages on sleep apnea surgery and uvulopalatopharyngoplasty cover the older options. For more depth on the Inspire device specifically, see the dedicated Inspire treatment page. If your CPAP is the problem, alternative treatments for sleep apnea and CPAP vs BiPAP might help. And if central events are part of your picture, central sleep apnea covers why HNS is not the answer there.
For patient-level information from sources I trust, the American Academy of Sleep Medicine and the Sleep Foundation are both good. The original STAR trial in the New England Journal of Medicine is publicly available if you want to read the foundational paper yourself.
⚠️ MEDICAL DISCLAIMER This blog provides general information only and is not a substitute for professional medical advice, diagnosis, or treatment. Sleep apnea is a serious condition, and CPAP equipment should be used under proper medical supervision. Always consult your doctor or sleep specialist before starting, stopping, or changing any therapy. I share personal experiences as a CPAP user, not as a medical professional. Individual results vary. For medical guidance, please consult a qualified clinician or the American Academy of Sleep Medicine (aasm.org).